In-Check™ DIAL G16
Inhaler Technique Training
and Assessment Tool
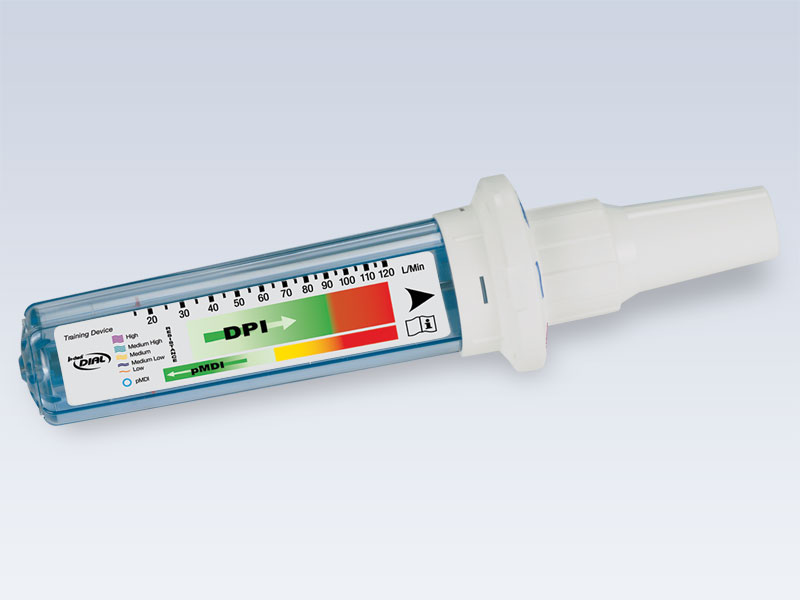
The In-Check DIAL is a hand held inspiratory flow measurement device with a dial top. The DIAL orifices have been designed to simulate the resistance of inhaler devices from the DPI and MDI categories. It enables clinicians to train patients to use more or less inspiratory force, to achieve their optimal flow rate with a particular MDI or DPI device.
Goal: Better lung deposition with less medication waste.
The DIAL can be adjusted to accurately simulate the resistance of popular inhaler devices which include MDI’s and DPI’s such as Turbuhaler®, Flexhaler®, Twisthaler®, Aerolizer®, Handihaler®, Diskus®, Ellipta® and Pressair® among others. The InCheck DIAL enables clinicians to train patients to the proper inspiratory technique considering force and flow rate to achieve improved understanding and ultimately better deposition of the medication being inhaled into the lungs.
The DIAL G16 now categorizes inhalers by resistance and the newly designed scale gives proper feedback on all efforts.
The one-way valved inspiratory mouthpiece allows for multiple patient use and is adaptable for adult or pediatric use protecting both the instrument and the patient from cross-contamination.
* Custom resistance adapters are available by calling Alliance Tech Medical.
With the In-Check DIAL you can quickly identify whether your patient has the correct technique for proper use of their inhaler.
- Ideal for children or adults having difficulty understanding proper technique
- Identifies the inhaler type. the resistance and corresponding optimal flow rate
- Trains patients to achieve optimal flow rates in a matter of minutes
- The one-way valved mouthpiece protects both the dial and the next patient
- One-way valved mouthpiece is adaptable for adult or pediatric use
Multiple Patient Use
Click photos to enlarge
Option 1:
Disposable Mouthpiece
One-way valve inspiratory mouthpieces help reduce risk of cross-infection to protect patient and equipment from contamination by preventing blowback.
- Order Number 3579912
Option 2:
Filtered Mouthpiece
Two way filter provides protection against possible cross-infection helping to protect patient and equipment. 99.9% Bacterial/Viral effective.
- Order Number 3579911
Accuracy
Every In-Check™ DIAL is individually calibrated to ensure a high degree of accuracy. Inspiratory flows in the range of 15 L/min to 120 L/ min can be measured. The In-Check Dial is accurate to +/- 10% or 10 L/min (whichever is greater). The In-Check DIAL has been tested by AEA Technology P.L.C. in the U.K. for accuracy, and has been verified as providing similar resistances to inhaler devices. The In-Check DIAL has been calibrated using an ATS pulmonary waveform generator. In-Check DIAL complies with the AS/NZS standard for back pressure in flow meters. Recommended Product Life is 2 years.
Cleaning
The In-Check DIAL should be cleaned by washing in warm soapy water, rinsed and air dried when used personally. See instructions for use for other sanitizing methods for multiple users.
Ordering Information
Education/Clinicals
Introduction
Inhalers and Inspiratory Flow
In-Check DIAL
The In-Check Dial is a low-range inspiratory flow meter (15 to 120 L/min) that assists the health care professional to assess inhaler technique and teach optimal inhaler use. It can simulate the different “internal resistance” for commonly used inhaler devices, enabling measurement of the speed of inhalation for each device chosen.
Readings of inspiratory flow rate obtained enable the healthcare professional to encourage patients to modify their inspiratory technique (by inhaling with more, or less effort), in order to achieve “optimum inspiratory flow”.
In-Check DIAL simulates resistance and measures flow
Resistance: The patient feels the same resistance as the real inhaler.
Flow: An accurate measurement of the speed of inhalation.
These two principles aid in inhaler training and assessment.
Performance Accuracy
Accuracy+/- 10% or 10 L/min (whichever is greater) and repeatability of +/- 5 L/min.
How to Reset the In-Check DIAL
- Hold the instrument vertically with the mouthpiece uppermost, so that the rounded end of the meter can be tapped against the other hand or a horizontal surface, such as a table.
- A gentle tap will dislodge the magnetic resetting weight, which will return the red cursor to a start position. When this has happened, the meter must turn through 180 degrees to return the magnetic weight to its resting position.
How to use the In-Check DIAL
Important: As with any inhalation device, it is important to check for loose foreign objects before the device is used. The transparent material used in the construction of the In-Check DIAL enables the user to make a visual check before inhalation.
- Reset the In-Check DIAL.
- Align the scale with the desired inhaler device – an audible “click” should be heard.
- Attach a clean mouthpiece (small mouthpieces can be used with the supplied adaptor).
- Ask the patient to exhale slowly and deeply.
- Seal lips around the mouthpiece. According to the inhaler chosen, instruct the patient to inhale in the manner recommended by the manufacturer.
- Record the inspiratory flow from the position of the red cursor against the scale. Reset, and repeat two more times.
- Compare values achieved with target flows for that device. To operate an inhaler device optimally, the patient should be able to achieve a flow rate within the Optimal Range. If other inhalers are used, then repeat steps 1 to 7.
Cleaning
Instructions
- Immerse In-Check DIAL in warm (but not hot) mild detergent solution for 2-3 minutes (maximum 5 minutes. Agitate the meter to ensure proper cleaning.
- Rinse in warm water and shake to remove any excess water. It is important to rinse thoroughly.
- Allow to dry completely before using again.
The expected life of the In-Check DIAL, with normal use, is two years.
In-Check DIAL
Peak Inspiratory Flow Meter
Speed of Inhalation Affects Several Parameters
The inspiratory flow through an inhaler is one of the factors that will affect the drug delivery from that device. A change in the speed of the air through a particular inhaler will influence some or all of the following:
Aerosol Quantity – total emitted dose for inhalation
Aerosol Quality – average particle size
Dose Consistency – total dose emitted over life of inhaler
Lung Deposition – total dose delivered
Side-Effect Risk – deposition in the mouth, throat and oropharynx
The result is that for some patients, changes in inspiratory flow will be associated with variations in clinical effect – pulmonary function parameters and relief from symptoms.
Optimum Inspiratory Flow Range
The most effective drug delivery occurs when the patient achieves a flow within the Optimum Inspiratory Flow Range.
At flow rates outside this optimum range, certain devices may deliver an effective (rather than optimum) dose. However, inspiratory flow rates outside the optimum range may reduce pulmonary deposition significantly.
By measuring the inspiratory flow of a patient using an inhaler, a comparison with the flow rates associated with most desirable performance and clinical effect can be made. This can help to identify those who are using their inhaler sub-optimally.
Inhaler Devices and Drug Delivery
Drug delivery from the various types of inhaler device is produced by different methods.
All inhaler devices are designed to deliver drug particles of a certain size to the small airways during inhalation. Particles of this size (generally agreed to be approximately between 2 and 5 microns) are known as the “respirable fraction”. The particles are either in aerosol (in a suspension or a solution) or dry powder form.
As the great majority of asthma medication is delivered via inhalers, optimum inhaler technique is an important factor in the management of this disease. Patients require their medication for both short-term relief and long term prevention, and the delivery of these drugs to the lungs is affected by inspriatory flow.
Common pMDI Metered Dose Inhaler, and MDI spacers with low resistance (e.g. AbleSpacer): 30 – 60 L/min
Autohaler® Automatic pMDI: 30 – 60 L/min<
Easibreathe Automatic pMD: 20 – 60 L/min
Diskus® Multiple-dose powder inhaler: 30 – 90 L/min
Turbuhaler® Turbulent flow inhaler: 60 – 90 L/min
Metered dose inhalers (pMDI)
With most MDI’s, the aerosol is delivered under pressure at high speed (often over 90 kilometers per hour). The inhalation should be slow and steady. Inhaling too fast may cause a greater proportion of the aerosol to impact at the back of the throat and be subsequently swallowed, thus reducing the beneficial clinical effect and increasing the potential for local and systemic side effects. With the majority of MDI’s, manual depression of the drug canister actuates the drug delivery. However, with breath-actuated metered dose inhalers (e.g. Autohaler®) the aerosol is released by mechanical actuation, triggered by the patient breathing in through the device at any flow rate above a minimum level. At inspiratory flows below this, the patient will not receive any of the medication, because a dose will not be released.
Metered dose inhaler with spacer
It is recognized that the optimum inhalation technique for using and MSI with holding chamber/spacer is a slow inhalation (30 to 60/min). As the resistance of the valves of most chamber/spacer devices are low, the In-Check DIAL can be set to ‘low resistance pMDI’ or “Freeflow” to provide an approximate resistance for inspiratory flow measurements to be made.
Dry powder inhalers (DPI)
Drug delivery from DPIs is triggered by inhaling through the device. A metered quantity of powder is drawn into the airflow, and follows a specific route within the inhaler, which creates a resistance that is designed to break up the medication into particles of a respirable size.
As the internal design of each DPI is different, and the formulation of medication is not identical, the resistance the patient encounters when inhaling – and the speed of inhalation at which the optimum performance occurs – will be different from device to device.
The effort required for the patient to achieve a given inspiratory flow will increase as the internal resistance of the device increases. For the same patient effort, the higher the resistance the lower the resulting inspiratory flow.
Bibliography In-Check Drug Delivery
Further Infomation:
Additional articles, reports, videos, and more are available on CD.
Contact your local representative
PO Box 6024
Granbury, TX 76049
1.800.848.8923 / 817.326.6357
Fax: 817.326.2182
© 2024 Alliance Tech Medical